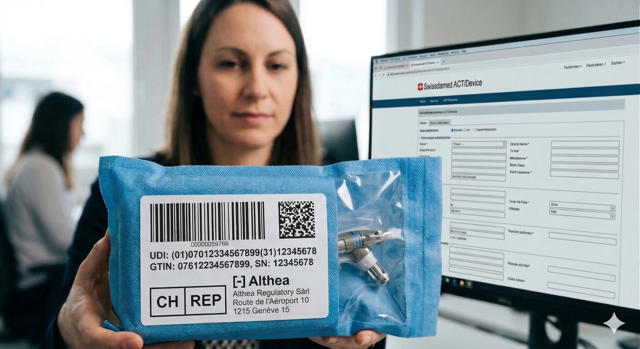
Swissmedic Establishment License
We offer full strategic and operational support to facilitate the successful introduction of medicinal products, including Pharmaceuticals, Medical Devices, Software as Medical Devices, IVDs, and Companion Diagnostics products, into the Swiss market.
We handle all aspects of the regulatory process, from establishing a local affiliate to building and maintaining Swiss Quality Management Systems (QMS) and overseeing post-approval licensing and maintenance (LCM – Life cycle management).
We support you in securing your Pharmaceutical Establishment License (PEL) and ensure strict compliance with Swissmedic regulations.
Helix Life Sciences provides Responsible Person (FvP/QP) and deputy services in full compliance with Swiss law, as well as acting as the local Responsible Person for pharmacovigilance (QPPV). We also ensure seamless regulatory compliance by preparing, attending, and following up on Swissmedic inspections on behalf of our clients.

- Our Capabilities
Accelerate Your Business Development Plans
Legal and Regulatory Expertise
Helix Life Science provides expert representation and guidance to foreign companies navigating Swiss legal and regulatory requirements, ensuring seamless compliance and market access.
Streamlined Submissions
Helix Life Science serves as the authorized applicant for Swiss Marketing Authorization Applications (MAAs), taking full responsibility for handling the entire regulatory submission process, from start to finish, for medical product manufacturers.
Trusted Liaison
Helix Life Science acts as a liaison between foreign companies and Swiss regulatory authorities, including SWISSMEDIC and FOPH/BAG, facilitating communication and ensuring seamless collaboration throughout the Swiss market access process.
- Market Presence
Tailored plans to establish your pharmaceutical business in Switzerland
Seamlessly enter the Swiss market with our expert support, ensuring regulatory compliance and product conformity.
Navigate complex Swiss regulations efficiently, as our team handles all aspects from local affiliate establishment, MAA submissions to ad hoc Swissmedic authorizations.
Establish a reputable brand in Switzerland, gaining recognition and trust among healthcare professionals and consumers.

Regulatory Experts
We provide expert representation on behalf of foreign companies before Swiss regional and cantonal authorities, ensuring seamless compliance with local regulations. We also act as liaison between foreign companies and Swiss federal authorities, primarily Swissmedic (Swiss Medicines Agency) and FOPH/BAG (Swiss Health Technology Assessment body). Furthermore, we serve as the authorized applicant for Swiss Marketing Authorization Applications (MAAs), handling the entire regulatory submission process from start to finish.
Our team of experienced regulatory experts possesses a deep understanding of Swiss regulatory requirements and a proven track record of success in guiding medical product manufacturers through the complex Swiss market access process. We are committed to providing our clients with personalized and tailored support to ensure the timely and successful introduction of their products into the Swiss market.
Start Your Journey with Helix
Drop your contact details into the form, and we’ll reach out to you!